PHARMACEUTICAL MANUFACTURING
The 22,500 m2 Plant of Saokim Pharma for the pharmaceutical production was built in Quang Minh Industrial Zone (Me Linh district, Hanoi, Vietnam) following the recommendations of the World Health Organization (WHO), including GMP/GLP/GSP-WHO certification and applying the ISO 9001:2000 quality management system.
CAPACITY OF FACTORY
· Formulations are being produced: film coated tablets, suppositories, hard capsules, soft capsules, pellets, drug release form at the target site, drugs for sprayed and dried such as granulates, powders, ect
· Capacity: 1 billion tablets/year
· New dosage forms will be available from 2017 onwards: liberated tablets, dispersible tablets, injectables, ect
TECHNOLOGIES
Saokim Pharma Factory applies the state-of-the-art technology and processes to date. Drugs are produced on a closed line, automatic, hygienic according to C.I.P. principle, production management by SCADA network with objective value, reliability in quality management.
API AND RAW MATERIAL MANUFACTURING
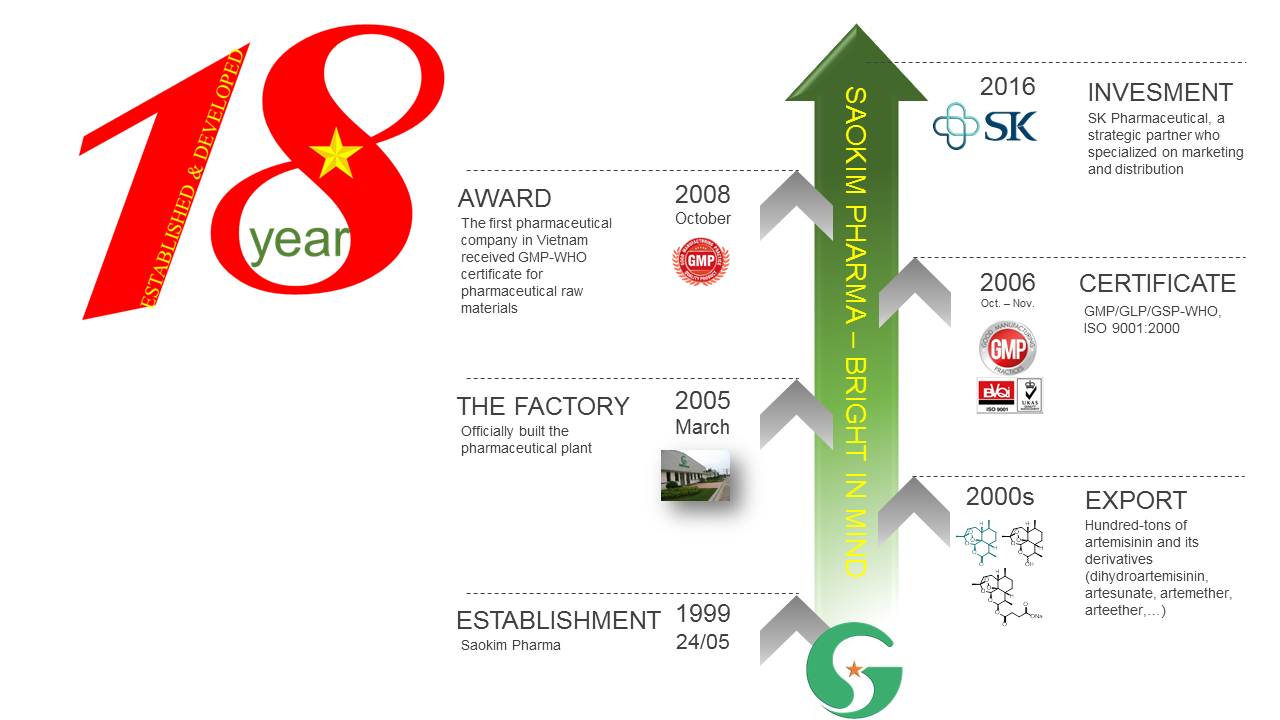
The Extracting and Isolating Factory produced the API and other raw materials, and the Semi-Synthesis Factory with an area of 11,000 m2 officially came into operation in 1999. The factory has a capacity of 100 tons of artemisinin, 30 tons of artesunate, 25 tons of artemether per year, ect
In October 2008, the factory was honored to be the first and only factory in Vietnam which was awarded GMP-WHO for the raw material production by the Ministry of Health of Vietnam, recommended by WHO.
With the capacity and prestige that has been established since its inception, Saokim Pharma has quickly become one of the largest producers of raw materials in the world. So far, the company has exported hundreds of tons of raw materials for antimalarial production to more than 30 countries in continent of Europe, Asia and Africa, including several multinational pharmaceutical corporations.